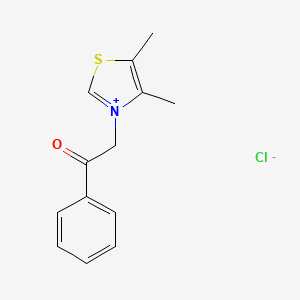
Alagebrium Chloride (ALT-711) – (341028-37-3)
Alagebrium chloride, also known as ALT-711, is a drug candidate that was developed by Alteon, Inc. It was the first compound to undergo clinical testing for its ability to break advanced glycation end-product (AGE) crosslinks, which are implicated in the aging process and various degenerative diseases. Alagebrium is designed to reverse the stiffening of blood vessel walls, a key factor in hypertension and cardiovascular disease, by targeting the biochemical pathways that lead to AGE formation.
The drug has shown efficacy in reducing systolic blood pressure and providing therapeutic benefits for patients with diastolic heart failure. In clinical trials, alagebrium demonstrated a decrease in left ventricular mass and improvements in left ventricular diastolic filling and quality of life in patients with diastolic heart failure. Additionally, it has been found to reduce renal mass and hyperfiltration in diabetic mice, indicating potential benefits for kidney function.
Despite its promising initial results, the development of alagebrium has faced challenges. Alteon merged with HaptoGuard in 2006, and the merged entity, Synvista Therapeutics, discontinued clinical trials of alagebrium in 2009 to focus on other clinical candidates and diagnostic tests. As a result, the future of alagebrium as a therapeutic agent remains uncertain.
The above information is displayed for information purpose only, and has not been reviewed by EON nor does EON attests or validates the accuracy nor does it constitutes a recommendation or validation.
https://en.wikipedia.org/wiki/Alagebrium
| Other Names | ALT-711 |
|---|---|
| IUPAC Name | 2-(4, 5-dimethyl-1, 3-thiazol-3-ium-3-yl)-1-phenylethanone;chloride |
| CAS | 341028-37-3 |
| Molecular Weight | 267.77 |
| Molecular Formula | C13H14ClNOS |
| SMILES | CC1=C(SC=[N+]1CC(=O)C2=CC=CC=C2)C.[Cl-] |