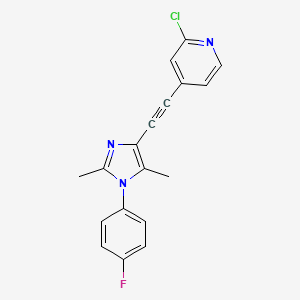
Basimglurant – (802906-73-6)
Basimglurant, also known as RG7090 and RO4917523, is a potent, selective, and orally available negative allosteric modulator of the metabotropic glutamate receptor 5 (mGlu5). Developed by Roche in collaboration with Chugai Pharmaceutical, it has undergone extensive preclinical and clinical research for its potential in treating treatment-resistant depression and fragile X syndrome.
In clinical trials, Basimglurant has demonstrated promising results. Phase I trials, completed in 2015, confirmed its safety and paved the way for Phase II studies.
Basimglurant’s drug-like properties are noteworthy. It has good oral bioavailability, a long half-life, and excellent brain penetration, supporting its potential for once-daily administration. Additionally, it exhibits antidepressant properties, as evidenced by its functional magnetic resonance imaging (fMRI) profile, and shows anxiolytic-like and antinociceptive features. These characteristics suggest it could address comorbidities such as anxiety, pain, daytime sleepiness, and apathy or lethargy associated with major depressive disorder.
Despite setbacks in its application for fragile X syndrome, Basimglurant remains a subject of interest for its potential in depression treatment. Its unique mechanism of action and favorable safety profile continue to drive research and development efforts, making it a promising candidate for future therapeutic applications in psychiatric disorders.
The above information is displayed for information purpose only, and has not been reviewed by EON nor does EON attests or validates the accuracy nor does it constitutes a recommendation or validation.
In clinical trials, Basimglurant has demonstrated promising results. Phase I trials, completed in 2015, confirmed its safety and paved the way for Phase II studies.
https://en.wikipedia.org/wiki/Basimglurant
| Other Names | RG7090 |
|---|---|
| IUPAC Name | 2-chloro-4-[2-[1-(4-fluorophenyl)-2, 5-dimethylimidazol-4-yl]ethynyl]pyridine |
| CAS | 802906-73-6 |
| Molecular Weight | 325.8 |
| Molecular Formula | C18H13ClFN3 |
| SMILES | CC1=C(N=C(N1C2=CC=C(C=C2)F)C)C#CC3=CC(=NC=C3)Cl |