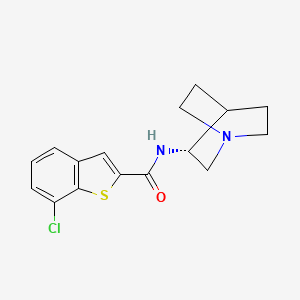
Encenicline – (550999-75-2)
Encenicline (EVP-6124) is a selective partial agonist of the α7 nicotinic acetylcholine receptor (nAChR). It was developed for the treatment of cognitive impairment in conditions such as schizophrenia and Alzheimer’s disease.
Encenicline underwent Phase III clinical trials but failed to meet study endpoints in 2016 due to rare but serious gastrointestinal side effects observed in patients.
The drug was being developed by FORUM Pharmaceuticals, which suspended all studies in 2015 pending further investigation. Encenicline showed promise in earlier trials at lower doses (2 mg/day), but the Phase III trials used a higher dose (3 mg/day), which may have contributed to the adverse effects. The drug’s development remains on hold as of recent updates.
The above information is displayed for information purpose only, and has not been reviewed by EON nor does EON attests or validates the accuracy nor does it constitutes a recommendation or validation.
https://en.wikipedia.org/wiki/Encenicline
| Other Names | EVP-6124, EVP 6124, UNII-5FI5376A0X |
|---|---|
| IUPAC Name | N-[(3R)-1-azabicyclo[2.2.2]octan-3-yl]-7-chloro-1-benzothiophene-2-carboxamide |
| CAS | 550999-75-2 |
| Molecular Weight | 320.8 |
| Molecular Formula | C16H17ClN2OS |
| SMILES | C1CN2CCC1[C@H](C2)NC(=O)C3=CC4=C(S3)C(=CC=C4)Cl |