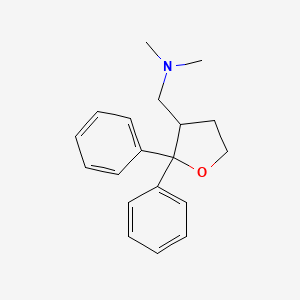
Blarcamesine – (195615-83-9)
Blarcamesine is an experimental drug under development for the treatment of Alzheimer’s disease and a variety of other neurological conditions. It acts as an agonist of the sigma σ1 receptor, the muscarinic acetylcholine M1 receptor, and the ionotropic glutamate NMDA receptor. Developed by Anavex Life Sciences, blarcamesine has shown promise in preclinical and clinical studies for its potential to improve cognitive function and protect against neurodegeneration.
In trials for Alzheimer’s disease, blarcamesine has demonstrated improvements in cognition and activities of daily living in patients with a fully functional SIGMAR1 gene, which encodes the σ1 receptor. The drug has also shown significant benefits in episodic memory in Parkinson’s disease patients after 14 weeks of treatment. Additionally, blarcamesine has been investigated for its potential in treating fragile X syndrome, Rett syndrome, and other neurological disorders, although development for some indications has been discontinued.
Blarcamesine’s mechanism of action involves modulating multiple pathways associated with neuroprotection and cognitive enhancement. It prevents the formation of β-amyloid plaques and tau hyperphosphorylation, which are hallmarks of Alzheimer’s disease. The drug also exhibits anti-apoptotic and anti-oxidant properties, contributing to its neuroprotective effects.
The above information is displayed for information purpose only, and has not been reviewed by EON nor does EON attests or validates the accuracy nor does it constitutes a recommendation or validation.
https://en.wikipedia.org/wiki/Blarcamesine
| Other Names | AVex-73, anavex2-73 |
|---|---|
| CAS | 1-(2, 2-diphenyloxolan-3-yl)-N, N-dimethylmethanamine |
| Molecular Weight | 195615-83-9 |
| Molecular Formula | 281.4 |