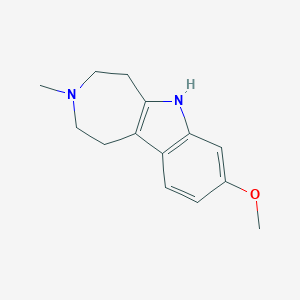
Tabernanthalog – (2483829-58-7)
Tabernanthalog is a novel water-soluble, non-toxic azepinoindole analog of the psychoactive drug Tabernanthine, first synthesized by Professor David E. Olson at UC Davis. Unlike its parent compound, Tabernanthalog is a non-hallucinogenic serotonin 5-HT2A receptor agonist and a serotonin 5-HT2B receptor antagonist, with high selectivity for serotonin 5-HT2 receptors. It also interacts with monoamine oxidase A (MAO-A), the α2A-adrenergic receptor, and the serotonin transporter (SERT).
In rodent studies, Tabernanthalog has shown promise in promoting structural neural plasticity, reducing drug-seeking behavior, and producing antidepressant-like effects. It effectively reduces motivation for heroin and alcohol in rats, indicating its potential in treating polydrug use disorders. As a member of the class of compounds known as non-hallucinogenic psychoplastogens, Tabernanthalog is being developed by Delix Therapeutics as a potential medicine for neuropsychiatric disorders.
The above information is displayed for information purpose only, and has not been reviewed by EON nor does EON attests or validates the accuracy nor does it constitutes a recommendation or validation.
https://en.wikipedia.org/wiki/Tabernanthalog
| Other Names | GTPL13551 |
|---|---|
| IUPAC Name | 8-methoxy-3-methyl-2, 4, 5, 6-tetrahydro-1H-azepino[4, 5-b]indole |
| CAS | 2483829-58-7 |
| Molecular Weight | 230.31 |
| Molecular Formula | C14H18N2O |
| SMILES | CN1CCC2=C(CC1)NC3=C2C=CC(=C3)OC |