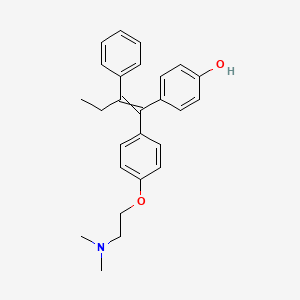
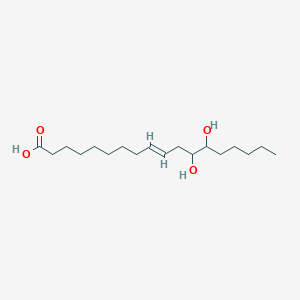
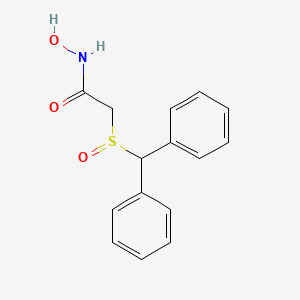
ABBV-CLS-7262 – (2945073-88-9)
Fosigotifator is an experimental small-molecule drug developed by AbbVie, currently undergoing clinical trials to evaluate its efficacy in treating amyotrophic lateral sclerosis (ALS).
It is formulated as a monosodium phosphate salt mixed with tromethamine, known as ABBV-CLS-7262. Fosigotifator acts as a prodrug targeting the modulation of the integrated stress response (ISR) pathway.
Fosigotifator sodium tromethamine is in Phase II clinical trials and is being investigated for its potential to inhibit ISR-related diseases.
The above information is displayed for information purpose only, and has not been reviewed by EON nor does EON attests or validates the accuracy nor does it constitutes a recommendation or validation.
https://en.wikipedia.org/wiki/Fosigotifator
| Other Names | Fosigotifator sodium tromethamine, BN9FN5629V, CHEMBL5314908, EX-A7829 |
|---|---|
| IUPAC Name | sodium;2-amino-2-(hydroxymethyl)propane-1, 3-diol;[(2S)-1, 4-bis[[2-(4-chloro-3-fluorophenoxy)acetyl]amino]-2-bicyclo[2.2.2]octanyl]oxymethyl hydrogen phosphate |
| CAS | 2945073-88-9 |
| Molecular Weight | 782.5 |
| Molecular Formula | C29H37Cl2F2N3NaO12P |
| SMILES | C1CC2(CCC1(C[C@@H]2OCOP(=O)(O)[O-])NC(=O)COC3=CC(=C(C=C3)Cl)F)NC(=O)COC4=CC(=C(C=C4)Cl)F.C(C(CO)(CO)N)O.[Na+] |