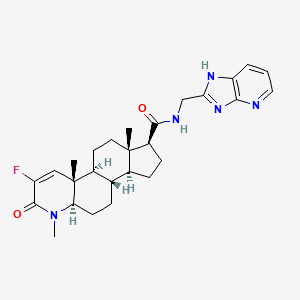
MK-0773 – (606101-58-0)
MK-0773 is a selective androgen receptor modulator (SARM) developed by Merck and GTx for the treatment of sarcopenia, a condition characterized by the loss of muscle mass and strength in older adults. This orally active compound is a 4-azasteroid and a potent agonist of the androgen receptor (AR). MK-0773 exhibits tissue-selective androgenic effects, significantly increasing lean body mass with anabolic effects comparable to approximately 80% of those induced by dihydrotestosterone (DHT). However, it demonstrates minimal impact on reproductive tissues such as the uterus, sebaceous glands, and prostate, indicating a reduced propensity for virilization.
In clinical studies, MK-0773 has shown promising results in elderly women with sarcopenia, leading to a significant increase in lean body mass and muscle strength compared to placebo, although the latter did not reach statistical significance. The drug also demonstrated a favorable safety profile, with no evidence of androgenization. However, some participants experienced elevated liver enzymes, which resolved after discontinuing the treatment.
The above information is displayed for information purpose only, and has not been reviewed by EON nor does EON attests or validates the accuracy nor does it constitutes a recommendation or validation.
https://en.wikipedia.org/wiki/MK-0773
| Other Names | PF-05314882, MK 0773, CHEMBL3221237 |
|---|---|
| IUPAC Name | (1S, 3aS, 3bS, 5aR, 9aS, 9bS, 11aS)-8-fluoro-N-(1H-imidazo[4, 5-b]pyridin-2-ylmethyl)-6, 9a, 11a-trimethyl-7-oxo-2, 3, 3a, 3b, 4, 5, 5a, 9b, 10, 11-decahydro-1H-indeno[5, 4-f]quinoline-1-carboxamide |
| CAS | 606101-58-0 |
| Molecular Weight | 479.6 |
| Molecular Formula | C27H34FN5O2 |
| SMILES | C[C@]12CC[C@H]3[C@H]([C@@H]1CC[C@@H]2C(=O)NCC4=NC5=C(N4)C=CC=N5)CC[C@@H]6[C@@]3(C=C(C(=O)N6C)F)C |