OPK-88004 – (1029692-15-6)
OPK-88004 is a non-steroidal selective androgen receptor modulator (SARM) developed by OPKO Health for the treatment of erectile dysfunction and symptoms associated with benign prostate hyperplasia. This indole derivative has been investigated for its potential to improve muscle mass and strength, bone formation, and erectile function without stimulating prostate growth.
In clinical trials, OPK-88004 demonstrated a dose-related increase in lean body mass and a decrease in body fat percentage. However, it did not show significant improvements in sexual function or physical performance. The compound was evaluated in a phase II trial for benign prostatic hyperplasia, but the study was terminated due to challenges in measuring prostate size.
Despite its potential benefits in increasing lean mass and reducing fat, OPK-88004 did not meet expectations for enhancing sexual symptoms or overall physical performance in men with prostate cancer. The drug is currently under investigation as an investigational new drug in the United States.
The above information is displayed for information purpose only, and has not been reviewed by EON nor does EON attests or validates the accuracy nor does it constitutes a recommendation or validation.
https://en.wikipedia.org/wiki/OPK-88004
| Other Names | LY2452473, LY-2452473, XKW9MYF94Y |
|---|---|
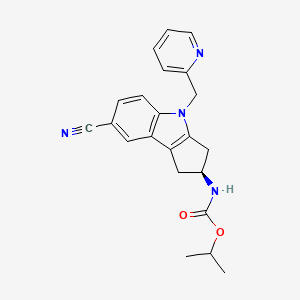
| IUPAC Name | propan-2-yl N-[(2S)-7-cyano-4-(pyridin-2-ylmethyl)-2, 3-dihydro-1H-cyclopenta[b]indol-2-yl]carbamate |
| CAS | 1029692-15-6 |
| Molecular Weight | 374.4 |
| Molecular Formula | C22H22N4O2 |
| SMILES | CC(C)OC(=O)N[C@H]1CC2=C(C1)N(C3=C2C=C(C=C3)C#N)CC4=CC=CC=N4 |