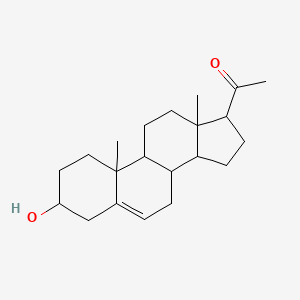
Pregnenolone – (145-13-1)
Pregnenolone is a crucial endogenous steroid and metabolic intermediate in the biosynthesis of various steroid hormones, including progestogens, androgens, estrogens, glucocorticoids, and mineralocorticoids. This neurosteroid is synthesized from cholesterol in the adrenal glands, gonads, and brain, primarily through the action of the enzyme cytochrome P450scc.
Pregnenolone plays a significant role in neuroprotection, enhancing synaptic function, and promoting myelination, which can lead to improvements in cognitive and memory functions. It acts as a negative allosteric modulator of the CB1 receptor, thereby preventing full activation by agonists like tetrahydrocannabinol (THC) and potentially protecting the brain from cannabis intoxication. Additionally, pregnenolone sulfate, a metabolite of pregnenolone, influences GABA and NMDA receptor activity and has been shown to activate the TRPM3 ion channel, influencing calcium entry and insulin release.
Research has demonstrated pregnenolone’s potential therapeutic applications, particularly in cognitive disorders and as a modulator of cannabinoid signaling. It has been investigated for its ability to mitigate the effects of THC and improve emotional regulation. Despite its promising properties, pregnenolone itself does not exhibit progestogenic, corticosteroid, estrogenic, androgenic, or antiandrogenic activities. Its role as a precursor and modulator of various hormonal pathways underscores its importance in endocrine and neurological health.
The above information is displayed for information purpose only, and has not been reviewed by EON nor does EON attests or validates the accuracy nor does it constitutes a recommendation or validation.
https://en.wikipedia.org/wiki/Pregnenolone
| Other Names | Arthenolone, Pregnetan |
|---|---|
| IUPAC Name | 1-[(3S, 8S, 9S, 10R, 13S, 14S, 17S)-3-hydroxy-10, 13-dimethyl-2, 3, 4, 7, 8, 9, 11, 12, 14, 15, 16, 17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl]ethanone |
| CAS | 145-13-1 |
| Molecular Weight | 316.5 |
| Molecular Formula | C21H32O2 |
| SMILES | CC(=O)[C@H]1CC[C@@H]2[C@@]1(CC[C@H]3[C@H]2CC=C4[C@@]3(CC[C@@H](C4)O)C)C |