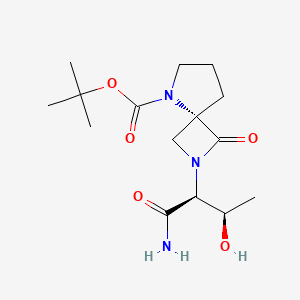
Zelquistinel – (2151842-64-5)
Zelquistinel is an investigational oral medication under development by Gate Neurosciences for the treatment of major depressive disorder (MDD). It is a small-molecule modulator of the N-methyl-D-aspartate (NMDA) receptor, acting through a unique binding site independent of the glycine site to enhance NMDAR-mediated synaptic plasticity.
Zelquistinel exhibits high oral bioavailability and dose-proportional exposures in plasma, brain, and cerebrospinal fluid (CSF). In preclinical studies, it demonstrated rapid and sustained antidepressant-like effects, enhancing long-term potentiation (LTP) in the hippocampus and medial prefrontal cortex (mPFC). The drug’s efficacy in rodent models of depression, such as the forced swim test and chronic social defeat model, supports its potential as a rapid-acting antidepressant.
In clinical trials, Zelquistinel has progressed to Phase IIb, following the completion of an exploratory Phase IIa trial by Allergan. The drug’s safety and tolerability are highlighted by its lack of sedative or ataxic side effects in rat models, distinguishing it from other NMDA receptor modulators like ketamine.
The above information is displayed for information purpose only, and has not been reviewed by EON nor does EON attests or validates the accuracy nor does it constitutes a recommendation or validation.
https://en.wikipedia.org/wiki/Zelquistinel
| IUPAC Name | tert-butyl (4S)-2-[(2S, 3R)-1-amino-3-hydroxy-1-oxobutan-2-yl]-3-oxo-2, 5-diazaspiro[3.4]octane-5-carboxylate |
|---|---|
| CAS | 2151842-64-5 |
| Molecular Weight | 327.38 |
| Molecular Formula | C15H25N3O5 |
| SMILES | C[C@H]([C@@H](C(=O)N)N1C[C@]2(C1=O)CCCN2C(=O)OC(C)(C)C)O |