- Home
- Products & Services
Check Out Our Research And Updates
You can consult the research papers, articles, and product release updates - Research
- Contacts
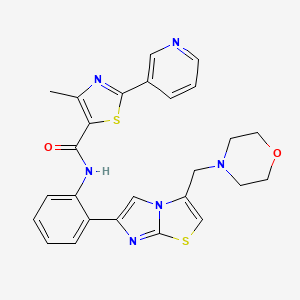
SRT-2104 – (1093403-33-8)
SRT-2104 is an experimental drug developed by Sirtris Pharmaceuticals as a selective activator of the sirtuin subtype SIRT1. Initially investigated for its potential in treating Type II diabetes, SRT-2104 progressed to Phase II clinical trials before development was halted.
Despite this, it remains a valuable tool in animal studies exploring SIRT1’s functions. SRT-2104 is a small molecule that increases SIRT1 protein levels without affecting SIRT1 mRNA, making it a unique and potent modulator of this enzyme. It has been studied for its effects on various conditions, including diabetes, Huntington’s disease, and sepsis, with research indicating its potential to reduce oxidative stress, improve motor function, and enhance survival in relevant animal models.
The drug is characterized by its favorable selectivity profile and good tolerability, with studies demonstrating a dose-dependent increase in exposure and a notable food effect that can be leveraged to optimize its pharmacokinetics. Although no longer in active clinical development, SRT-2104 continues to be a subject of interest in preclinical research, offering insights into the therapeutic potential of SIRT1 activation.
The above information is displayed for information purpose only, and has not been reviewed by EON nor does EON attests or validates the accuracy nor does it constitutes a recommendation or validation.
SKU:
1093403-33-8
Categories: Biochemical, Nootropic
https://en.wikipedia.org/wiki/SRT-2104
| Other Names | SRT 2104, SRT2104 |
|---|---|
| IUPAC Name | 4-methyl-N-[2-[3-(morpholin-4-ylmethyl)imidazo[2, 1-b][1, 3]thiazol-6-yl]phenyl]-2-pyridin-3-yl-1, 3-thiazole-5-carboxamide |
| CAS | 1093403-33-8 |
| Molecular Weight | 516.6 |
| Molecular Formula | C26H24N6O2S2 |
| SMILES | CC1=C(SC(=N1)C2=CN=CC=C2)C(=O)NC3=CC=CC=C3C4=CN5C(=CSC5=N4)CN6CCOCC6 |
Related products


Quick Contact
If you want to order or have any questions, feel free to contact our team.
Send Us Email:
Call Us Today (WhatsApp):
©2025 EON Biotech, All Rights Reserved.